A Risk-Based Case for Analytical Redundancy in the Pharmaceutical Lifecycle
Author’s Note (Important Context)
This article presents a proposed, proactive, risk-based analytical strategy based on professional experience and interpretation of current ICH and regulatory guidance. It is not a regulatory requirement and is intended only for complex and critical molecules. Any alternative analytical procedure must be scientifically justified, validated for its intended use, and approved by relevant regulatory authorities before routine application.
Author: Hemant Patil
Pharmaceutical Professional | AscentCoAI Pharma
LinkedIn: https://www.linkedin.com/in/hemant-patil-pharma/
LinkedIn: https://www.linkedin.com/company/1082213376/
1. Introduction: Why This Matters Now
In the modern pharmaceutical landscape, analytical methods are the bedrock of quality assurance. However, a single, static primary method presents inherent risks to drug supply and quality control over a product’s commercial lifetime. The following challenges underscore the need for proactive thinking:
- Supply Chain Vulnerabilities: Discontinuation of columns, filters, or critical reagents.
- Process & Supplier Changes: Alterations that impact chromatographic behavior or sample preparation.
- Material Source Changes: SUPAC-level changes (e.g., alternate API or excipient vendor) that can introduce new impurities or matrix interferences, challenging the specificity of the primary method.
- Operational Transfers: Site or instrument transfers revealing method robustness issues.
- Method Drift: Gradual performance changes over long commercial lifetimes.
- Evolving Impurity Profiles: Emergence of new impurities during stability not resolved by the primary method.
- Inconclusive Investigations: OOS/OOT results with no clear assignable cause linked to the primary method.
Reactive method redevelopment or remediation is costly, time-consuming, and threatens supply continuity.
2. The Proactive Concept: Clearly Defined
For high-risk molecules, develop one additional, orthogonal analytical method during R&D and integrate it into the analytical control strategy with predefined governance and decision rules. This is structured risk mitigation not duplication. The alternative method serves as a scientifically justified, validated tool for specific, predefined scenarios.
3. Regulatory Alignment
This strategy is grounded in and supported by modern regulatory and pharmacopeial frameworks:
- ICH Q14: Promotes risk-based analytical development, the Analytical Target Profile (ATP), and lifecycle management, justifying alternative methods as part of a robust control strategy.
- ICH Q2(R2): Emphasizes fitness-for-purpose validation. The alternative method is validated to a scope aligned with its specific, limited intended use.
- USP <1220> (Analytical Procedure Lifecycle): Advocates for procedure performance monitoring and having a science-based plan for method changes or investigations.
- Regulatory Precedent: Inspections value orthogonal confirmation for investigations and evidence of scientific, proactive quality management.
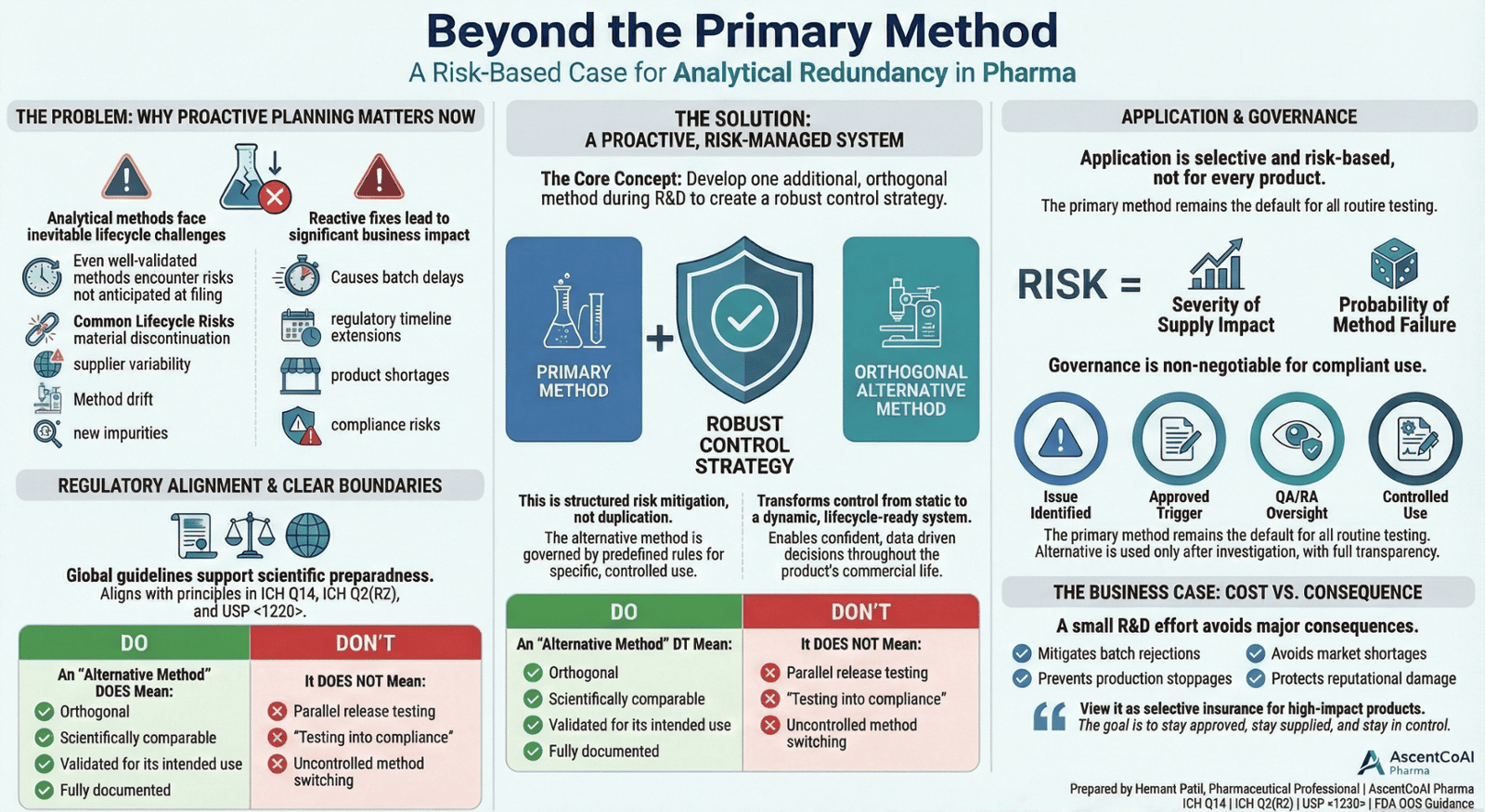
Figure 1: Overview of the risk-based analytical redundancy strategy - from problem identification to governance and business value
4. What an Alternative Method Means (and Does Not Mean)
It DOES mean:
- A scientifically comparable but orthogonal methodology (e.g., HPLC-UV vs. UPLC-DAD, or LC-MS for identification).
- Validated to a scope aligned with its intended, limited use (e.g., for impurity confirmation, not necessarily full release).
- Documented, approved, and controlled within the Pharmaceutical Quality System.
It DOES NOT mean:
- Parallel routine release testing.
- “Testing into compliance.”
- Uncontrolled method switching.
5. Selective Application – A Risk-Based Decision
Implementation is not universal. A risk assessment, grounded in the Analytical Target Profile (ATP) per ICH Q14, should guide application:
Risk Indicator = Severity of Supply Disruption × Probability of Method Failure
- High-Risk Candidates: Complex molecules (e.g., peptides, oligonucleotides), products with a history of method issues, sole-source therapies, or drugs with narrow therapeutic indices.
- Lower-Risk Candidates: Simple, well-understood molecules with highly robust and stable methods.
6. Practical Value Across the Lifecycle
- R&D & Stability: Provides improved impurity understanding and method specificity early on.
- Manufacturing & QC: Enables faster, science-based resolution of OOS/OOT investigations by ruling in/out analytical method causes.
- Business Continuity: Drastically reduces regulatory and supply disruption risk during method-related crises (e.g., column obsolescence).
7. Governance and Controlled Use
Clear governance is critical to prevent misuse and ensure regulatory compliance:
- The Primary Method remains the default for all routine testing.
- The Alternative Method is used only after a documented investigation and upon meeting predefined, approved triggers.
- Triggers (e.g., primary method failure, critical reagent discontinuation, unresolved OOS) are defined and approved by QA/Regulatory.
- All use of the alternative method is fully documented, tracked, and subject to change control.
8. Implementation
- SOPs: Establish an SOP for the management of alternative/orthogonal analytical methods.
- Validation: Define the validation scope (accuracy, precision, specificity, etc.) based strictly on the method’s intended contingency use.
- Clarity: Maintain a clear, auditable distinction between contingency use and routine use in all documentation.
9. Management Perspective
The incremental investment in developing and validating a contingency method during R&D is minimal compared to the potential costs of:
- Production stoppages.
- Prior Approval Supplement (PAS) submission timelines.
- Market shortages and lost revenue.
- Reputational damage from quality issues.
This is selective insurance for high-impact products, protecting patient supply and the company’s bottom line.
10. Final Takeaway
This strategy applies modern regulations intelligently and proactively. It moves the CMC framework from a static compliance record to a <b>resilient, adaptable, and lifecycle-ready system</b>. By planning for analytical uncertainty, we build greater certainty into drug supply.
Key References
- ICH Q14 – Analytical Procedure Development
- ICH Q2(R2) – Validation of Analytical Procedures
- USP <1220> – Analytical Procedure Lifecycle
- FDA Guidance for Industry: Investigating Out-of-Specification (OOS) Test Results for Pharmaceutical Production
- FDA Guidance for Industry: SUPAC-IR: Immediate Release Solid Oral Dosage Forms (1995)
- FDA Guidance for Industry: SUPAC-IR/MR: Manufacturing Equipment Addendum (2014)





